
CLINICAL SAFETY AND ACCEPTABILITY OF IOCIDE® VAGINAL GEL
JM Piper¹, P Siegel², B Bhatt2, J Morrison², and G Siegel²
¹University of Texas Health Science Center at San Antonio, Texas,
²Biomedical Development Corporation, San Antonio, Texas
Introduction
Iocide® is a novel, iodine-based, topical microbicide developed for vaginal use. Current clinical studies are focused on safety and acceptability of a single application to prevent intrapartum transmission of group B streptococcus (GBS) from mother to baby. Current strategies for prevention of early-onset GBS sepsis in the neonate require screening at 35-36 weeks and antibiotic administration during labor. The episodic nature of maternal GBS colonization and increasing antibiotic resistance make this approach suboptimal. Availability of a safe and effective microbicide for intravaginal use during labor would simplify prevention of this severe neonatal infection.
Objectives
To determine if Iocide® Vaginal Gel is safe for use as a single intravaginal dose in healthy nonpregnant women.
To determine if women find Iocide® Vaginal Gel
acceptable for this use.
Methods
Following FDA approval of the IND and IRB approval of the research protocol, Phase 1 human safety studies were initiated. Nonpregnant women proven free of vaginal lesions and sexually transmitted disease were included (those with STDs or vaginal lesions were treated and excluded from further participation).
Study Visits
1 Screening visit
(STD tests and exam for lesions)
2a Pretreatment exam and cultures
*** Iocide® vaginal gel treatment
2b 1 hour Post-treatment exam and cultures
3 Day 1 Post-treatment exam and cultures
4 Day 7 Post-treatment exam and cultures
Study Procedures
Testing Performed at Each Visit
- Hematology
- Serum Chemistry
- Urinalysis
- Thyroid Function Studies
- Vaginal Cultures
- Vaginal Inspection
Testing Performed at Specific Visits Only
STD testing (Visit 1)
Vaginal Biopsy (Visits 2a & 2b)
Product Satisfaction Survey (Visit 4)
Study Population:
16 women completed the study

Gynecologic Symptomatology
Impact on Bacterial Vaginosis Score
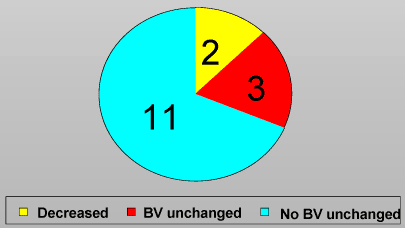
No subjects had an increased BV score
Product Acceptability
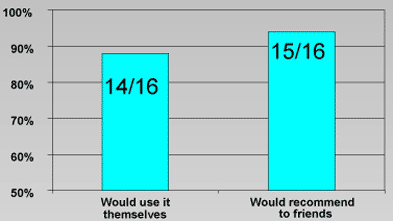
Results
Results of visual inspection:
2 women with mild vaginal inflammation at 24 hours, both resolved by final visit
Results of Safety Testing:
Hemotology, serum chemistry, urinalysis, and thyroid function results remained unchanged from baseline values
Conclusions
A single application of Iocide® Vaginal Gel in nonpregnant women does not significantly alter baseline values or vaginal appearance. Women exposed to the gel find it acceptable for personal use and would recommend it to their friends if available for vaginal cleansing.
Acknowledgements
Funding was provided to Biomedical Development Corporation by the National Institute of Child Health and Human Development – SBIR 5R44HD034990