SAFETY AND ANTIMICROBIAL EFFICACY OF EPIKEIA® TECHNOLOGY, AN ACETONE AND POLYMER-BASED SKIN COATING
NJ Girard¹, SE Piña², EK Eskew², WD Warren², BK Bhatt², and G Siegel²
¹University of Texas Health Science Center and ²Biomedical Development Corporation, San Antonio, TX
Introduction
Epikeia® is an acetone and polymer-based skin coating technology that has been developed as a combination barrier product and drug delivery vehicle. The technology has the potential to address numerous distinct clinical applications by incorporating active ingredients to create dermatologicals, active skin care products, and cosmeceuticals.
Some concern existed regarding the irritation potential of Epikeia® because acetone can be drying to skin. The studies described herein were performed on the basic vehicle (i.e., not containing any active ingredients), and demonstrate that Epikeia® is non-irritating to the skin of human subjects, and antimicrobial to numerous pathogens in vitro. These studies were completed under grants from the National Institutes of Health – SBIR No. 5R42AR44435.
21-Day Cumulative Irritation Study
OBJECTIVE: This study was performed to determine the irritation potential of Epikeia® skin coatings.
METHODS: The 21-day cumulative irritation study in humans is a standard FDA recommended test to determine the irritation potential of substances on human skin. Epikeia® coatings were applied to the backs of 26 human subjects over a 21 day period and scored for irritation. Every 24 hours, coatings were removed, skin was scored for erythema (0 – no erythema to 4 – severe erythema) and edema (0 – no edema to 4 – severe edema), and the coatings were reapplied.
RESULTS: No evidence of any erythema or edema was found on any subject at any time. There was never a score greater than 0 on any site during the 21 day study.
Antimicrobial Efficacy Study
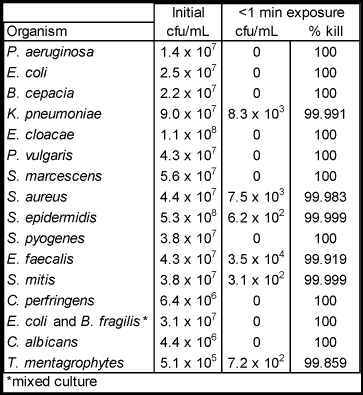
OBJECTIVE: This study was performed to evaluate the antimicrobial efficacy of Epikeia® skin coatings against a variety of pathogens commonly associated with surgical wound infections.
METHODS: Antimicrobial efficacy was evaluated in vitro against eight gram negative organisms, six gram positive organisms, and fungi. Microorganisms were grown to a concentration in the starting inoculum within 5 x 107 and 5 x 108 cfu/mL and exposed to either the skin coating solution or an acetone-only control. After an incubation period of 0, 0.5, 3, and 8 hours, microbial counts were taken.
RESULTS: Both the skin coating and the acetone-only test groups demonstrated a killing range of 99.859 – 100% at less than 1 minute. Both groups were effective at all time points indicating that acetone is responsible for antimicrobial activity.
Antimicrobial efficacy of Epikeia® against causative pathogens
Summary
Epikeia® skin coatings proved to be non-irritating to subjects in the 21-day Cumulative Irritation Study and demonstrated a seven log kill to assorted bacteria and fungi in less than one minute.
Conclusion
Despite having a relatively high concentration of acetone, Epikeia® skin coatings are nonirritating to human skin. The inclusion of acetone in the product enables Epikeia® to be antimicrobial to a wide range of pathogens.
Reference
United States Department of Health and Human Services. Food and Drug Administration, CDER. Guidance for Industry: Skin Irritation and Sensitization Testing of Generic Transdermal Drug Products, 1999.
Presented at the 27th Hawaii Dermatology Seminar February 14 – 22, 2003