SUPPRESSION OF CANDIDA BIOFILMS WITH AN IODINE ORAL RINSE
B.M. BHATT, C.J. GAUNTT, K.P. MCCLOSKEY, and G. SIEGEL
Biomedical Development Corporation, San Antonio, TX, USA
Abstract
Objective: The purpose of this experiment was to determine the inhibitory effectiveness of several Iocide Oral Rinse formulations (OR) in the metabolic activities of cells in Candida albicans biofilms as assessed by XTT assay.
Methods: Candida albicans (ATCC# SC5314) was propagated, washed 3X by PBS and suspended to 1×106 cells/ml in RPMI. One-tenth ml aliquots of the cell suspension were seeded to wells of 96 well microtiter plates. The plates were incubated for 24 or 48 hr at 37°C to allow biofilm to form, which was confirmed by microscopy at 40X. Unattached cells were removed by washing thrice with PBS and then 0.1ml PBS was added per well. Groups of 8 cell cultures were treated with 0.1ml of OR containing 0.012%, 0.045%, 0.068%, or 0.135% iodine, OR without Iodine (vehicle control), 0.12% Chlorhexidine Gluconate (CHX), Fluconazole (10mg/ml, oral suspension), or RPMI 1640 supplemented as above (growth control) for 30, 60 or 120 seconds. Sodium thiosulfate was added to each well containing OR to neutralize free iodine or 100 μl of PBS was added to biofilms receiving other treatments.
After removal of all fluids, the biofilms were washed twice with 0.1ml PBS and the XTT assay was performed. The metabolic activities in biofilm cells treated with OR formulations and Fungicides, were compared to that of the untreated biofilm cells.
Results: The reduction in metabolic activity due to OR formulations increased corresponding to the increasing level iodine and time of exposure. OR formulations containing 0.068% or 0.135% iodine were more effective in reducing metabolic activities than 0.12% chlorhexidine gluconate or fluconazole at 60 and 120 seconds of treatment. Metabolic activity of fluconazole actually decreased at the latter time points.
Conclusions: Oral Rinse formulations demonstrate a dose-duration-response in reducing metabolic activity of C. albicans biofilm and are more effective than Chlorhexidine gluconate or Fluconazole in inhibiting metabolic activities of 24/48 hour old C. albicans biofilms, as determined by the XTT assay.
Introduction
Over 1.4 million new cases of cancer will be diagnosed in 2007. These are typically treated with radiation therapy or chemotherapy. Radiation therapy or chemotherapy for cancer suppresses the immune system, which causes the patient to become more susceptible to oropharyngeal candidiasis.
Candida albicans has been recognized as the major etiologic microorganism of oropharyngeal candidiasis and/or fungemia in cancer patients for several years. Treatment of fungal infections in immunosuppressed cancer patients is standard practice, but antifungal therapy results in side effects of variable severity. In addition, isolates of some Candida are quite resistant to some antifungal drugs.
Iocide is a novel iodine-based antimicrobial oral rinse developed for the treatment of biofilm, specifically for the treatment of oral biofilm associated with candidiasis or gingivitis.
Methods
The metabolic activity of C. albicans cells in biofilms was determined by the XTT (2,3-bis(2- methoxy-4- nitro-5-sulfo-phenyl)-2H-tetrazolium- 5-carboxanilide) reduction assay, as described by Ramage. The XTT measures metabolic activity of mitochondrial dehydrogenases in live cells; these enzymes convert a colorless XTT tetrazolium salt into a formazan-colored product. XTT assay absorbance readings have been found to be proportional to the cellular density of the biofilm.
Candida albicans (ATCC# SC5314) was propagated in yeast peptone dextrose medium for 18-20hr at 37°C in an orbital shaker. After washing thrice with phosphate-buffered saline (PBS), the cells were suspended to 1×106 cells/ml in RPMI 1640 medium containing Lglutamine (2mM/ml) and morpholinepropanesulfonic acid. One-tenth ml aliquots of the cell suspension were seeded to wells of 96 well microtiter plates. The plates were incubated for 24 or 48 hr at 37°C to allow biofilm to form. Formation of biofilms was confirmed by microscopy at 40X. Unattached cells were removed by washing thrice with PBS and then 0.1ml PBS was added per well. Groups of 8 cell cultures were treated with 0.1ml of iodine-based oral rinse with 0.12%, 0.45%, 0.68%, or 0.135% iodine, the oral rinse lacking iodine (vehicle control), chlorhexidine gluconate oral rinse (CHG, 0.12%), fluconazole (Fluc, 10mg/ml, oral suspension), or RPMI 1640 supplemented as above (growth control) for 30, 60 or 120 sec. Sodium thiosulfate (25 μl of 0.1N) was added to each well containing OR with iodine to block further activity or 100 μl of PBS was added to biofilm cultures receiving other treatments. After removal of all fluids, the biofilm cultures were washed twice with 0.1ml PBS and the XTT assay
was performed as described by Ramage et al.
Results
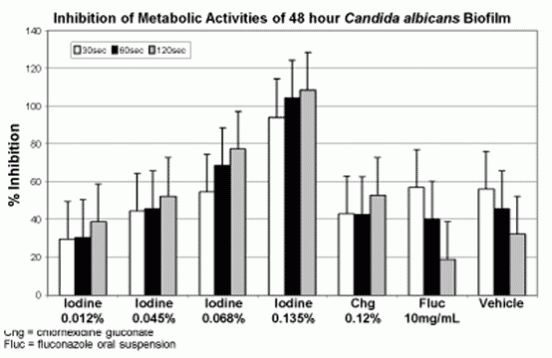
Data from 24 and 48hr C. albicans biofilms were similar; therefore only data from the 48hr old biofilm are presented in the Figure below. The results are presented as percent reduction of metabolic activities in biofilm cells by Iocide formulations compared to untreated control biofilm cells, as determined by XTT assay. Oral rinse formulations containing higher concentrations of iodine and longer times of treatment were more effective in inhibiting some metabolic activities of Candida albicans cells in biofilms. Oral rinse formulations containing 0.68%, or 0.135% iodine were more effective in reducing metabolic activities than 0.12% chlorhexidine gluconate or fluconazole at 60 and 120 sec of treatment.
Conclusions
Iodine-based oral rinse formulations are effective in vitro against C. albicans biofilm. The iodine formulations demonstrate a dosedependent response and the higher concentration formulations are more effective than chlorhexidine or fluconazole in the XTT assay.
Reference
Ramage G, K Vande Walle, BL Wickes, and JL Lopez-Ribot, Standardized method for in vitro antifungal susceptibility testing of Candida albicans biofilms. Antimicrob Agents Chemother, 2001. 45(9): 2475-9.
Acknowledgements
National Institutes of Health / National Institute of Dental and Craniofacial Research. SBIR# R43DE17301.
Frio Pharmaceuticals, San Antonio, TX
Presented at the: 4th ASM Conference on Biofilms March 25 – 29, 2007 Quebec City , Quebec , Canada