
TREATMENT TO IMPROVE SKIN TEXTURE (WRINKLING) IN HANDS
G Siegel¹, BM Bhatt¹, PB Siegel¹, HL Cardenas2, and HR Rawls²
¹Biomedical Development Corporation, San Antonio, TX, ²University of Texas Health Science Center at San Antonio,TX
Background
This study investigated the effectiveness of a novel polymer-based skin product to improve the texture of skin in hands. The product is an acetone and polymer-based skin coating technology that has been developed as a combination barrier product and drug delivery vehicle. The technology has the potential to address numerous distinct clinical applications by incorporating active ingredients to create dermatologicals, active skin care products, and cosmeceuticals.
The product, which contained tocopherol acetate (vitamin E), was applied overnight to the skin as a liquid that rapidly dries (~15 – 30 seconds) to form a microporous thin film. Previous subjective observations on a single individual indicated that daily treatment had a strong anti-wrinkle effect when applied to hands. This study was performed to confirm these initial results in a controlled trial.
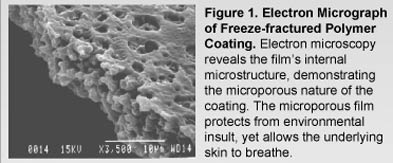
Study Subjects
Sixty-six women and one man completed the study. Study participants were given a brief oral presentation describing the background of the product, and informed consent was received prior to admission in the study. No specific exclusion or inclusion criteria were used, particularly with regard to age of the subject or appearance of wrinkles on hands. Subjects were given written and oral instructions on the use of the product.
Subjects were assigned sequential numbers for identification and random numbers for the before-treatment and after-treatment measurements. In this manner, all procedures and methods could be carried out in a doubleblind fashion.
Materials and Methods
Subjects were instructed to apply the product to their left hand each night before bedtime, and to remove the product the following morning. Skin impressions (as described below) were taken at baseline and after 3 months treatment. All profilometry data from the study subjects who completed the trial were included in the data set.
Skin Impressions. Replicas of the skin were made by taking an impression on the web of skin located between the thumb and forefinger of the left hand of each subject, using a low viscosity, hydrophilic dental impression material (Splash™). The impressions were made by holding a 10 mm diameter brass ring containing the impression material against the sampling site until it cured to a moderately hard rubber. Positive replicates were made from the negative impressions, using a low viscosity epoxy that was allowed to cure overnight
Surface Texture. A profilometer (Surtronic 3+™, Taylor Hobson Inc.) was used to determine surface texture of the epoxy replicates as shown in Figure 2. This instrument has a stylus, which travels across a surface and records vertical movements as a function of horizontal travel, measuring the up and down movements, and converting them to electrical signals. The electrical signals are used to record up and down travel (highs and lows) and distance between “peaks” and “valleys.” Five vertical parameters and one horizontal parameter were recorded.
Of the five vertical parameters, wrinkle depth (Ry) was measured as the average of the largest peak to valley heights among the three cut-off lengths that constituted each measurement. As a reasonable measure of spacing between wrinkles, the horizontal parameter, Sm, represents the average spacing between peaks in the 7.5 mm evaluation length traversed during a measurement. Thus increased values of Ry imply deeper wrinkles and increased values of Sm imply fewer (i.e., more widely spaced) wrinkles, and vice versa.

Figure 2. Surtronic 3+ Profilometer was used to measure texture of epoxy skin impressions.
Results
Data were analyzed using Student’s t-test for paired observations. Testing in two directions perpendicular to each other (0-deg and 90-deg) was completed with three replications in each direction for a total of six samples per subject per time point. Analyses reported are based on averages taken over all six samples at each time period.
As shown in Table 1, both outcomes showed significant changes from baseline to the end of treatment. As shown in Figure 3, 67% of subjects showed an increase (improvement) in wrinkle spacing (increased spacing = fewer wrinkles), and 33% showed no change or a decrease in spacing. The average subject showed a 19% improvement.

Table 1. Statistical analysis of skin texture measurements at baseline and end of treatment (n=67).
The greater the distance between peaks and valleys (Sm) and the smaller the peaks (Ry) are, the smoother the skin is. The average peak size changed by only 15 μm, or 10%, remaining at less than 200 μm. Although this is statistically significant, it is a very small change and unlikely to be perceptible. The spacing between peaks, which can be interpreted as the distance between wrinkles, also increased significantly (P = 0.015), from about 728 μm to an average of 828 μm, an increase of 100 μm, a readily detected change. The larger the spacing is between peaks, the fewer the wrinkles. Thus, this is indicative of an overall reduction in wrinkles of 19%. Of the 67% of subjects showing an improvement, the average reduction in wrinkle spacing was 39%. Improvements ranged up to 200%.
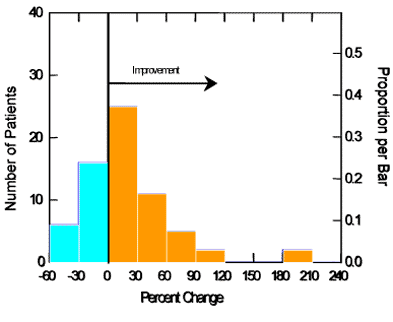
Figure 3. Subjects demonstrating various levels of change in skin wrinkling, based on increased spacing between peaks, Sm.
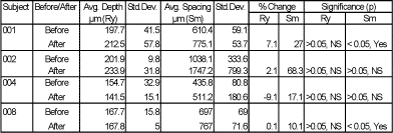
Table 2. Representative skin texture measurements at baseline and end of treatment.
Discussion
The mechanism responsible for the efficacy of the novel polymer coating in improving skin texture is not completely understood at this time. It is hypothesized that the product promotes improved skin barrier function by mediating transepidermal water loss (TEWL) at night, when TEWL is at its greatest. In addition, the product’s efficacy may be attributed to the topical delivery of Vitamin E or the mild exfoliation that occurs when the product is removed, all of which contribute to healthy skin.
Presented at: American Academy of Dermatology 63rd Annual Meeting. New Orleans, LA, USA. February 18-23, 2005. Poster# P227
Sponsored by Epikeia Incorporated