INCREASED BIOAVAILABILITY OF MICONAZOLE IN HUMAN SKIN WITH A NOVEL POLYMER VEHICLE
LK Pershing¹, JL Corlett¹, P Siegel² G Siegel²
Dept. Dermatology, Univ., of Utah, Salt Lake City, UT¹, and Biomedical Development Corp., San Antonio, TX²
Objective
The purpose of this study was to evaluate a novel polymer-based topical vehicle to deliver miconazole to human skin. The current study compared the dermato-pharmacokinetic uptake/elimination profiles of miconazole nitrate in a cream vehicle and miconazole base in the novel polymer delivery vehicle. To account for the differences in weight between the base and nitrate forms of miconazole, the drug concentrations were 1.738% miconazole base in the polymer vehicle and 2% miconazole nitrate in the cream.
Miconazole uptake into, and elimination from, the skin was quantified by harvesting the rate-limiting barrier, stratum corneum, with adhesive discs (a.k.a. skin stripping), then extracting the adhesive discs and quantifying drug content by High Performance Liquid Chromatography (HPLC).
Background
Miconazole nitrate cream, 2% is an approved over-the-counter product for the topical treatment of athlete’s foot, ringworm or jock itch caused by Trichophyton, Epidermophyton and Microsporum infections. The novel polymer vehicle is an acetone and polymer-based skin coating technology that has been developed as a combination barrier product and drug delivery vehicle. The product is applied to the skin as a liquid that rapidly dries (~15 – 30 seconds) to form a microporous thin film.
Effective topical drug therapy requires optimal drug concentrations be attained and maintained in skin to exert its pharmacological activity. This outermost layer of skin, the stratum corneum, is believed to be the “rate limiting barrier” to all drug absorption into and across the skin. If a topically applied drug does not partition into the stratum corneum, there will be no pharmacodynamic activity and thus, no effective therapy. Effective drug therapy also requires that the drug eliminate from the skin. Failure of the drug to eliminate from the skin may result in a toxic side effect.
Methods
Miconazole nitrate and base were quantified in stratum corneum samples harvested from treated skin sites using a validated HPLC assay (sensitivity limit 1 ng) following a single ~5 mg dose of each product (4.8 μl cream & 5.6 μl novel polymer vehicle) applied to each subject in the same study period. Ten sites (1.33 sq cm diameter) on each arm were randomly treated with both products for 0, 0.17, 0.75, 2.0, 4.0, 12 or 24 hours. Two skin sites above the forearm were used as untreated controls. Residual product was removed with 2 adhesive discs.
Stratum corneum was harvested from skin sites with a forceps and 12 additional adhesive discs (CuDerm Corp) at the end of application and 4, 8, 12, 24 hours after removal of a 24 hr application (elimination). Discs 1 and 2 of the 12 were discarded as possible drug contaminated and the remaining 10 discs combined for extraction and quantification by HPLC analysis.
Analysis
Peak concentration (Cmax) was the observed maximum value during the absorption period 0.0 to 24.0 hours. The time to peak concentration (Tmax) was the collection time at which Cmax was observed during the absorption period, 0.0 to 24.0 hrs. Area-under-the-drug content-time curve to the last measured concentration (AUC0-t) was calculated by the linear trapezoidal method. Cmax, Tmax, AUC0-t and Thalf were statistically evaluated for each product using pairwise-t test on means using Statview® (Abacus Concepts, Calabassas, CA, version 4.1).
Results
Six subjects, aged 38±4 yrs, completed the trial. All of the available data were used in the pharmacokinetic analyses. As shown in Table 1, the novel polymer vehicle produced 3.6-fold greater mean Cmax and AUC0-t of miconazole base than the cream (6.53 vs. 1.82 μg/sq cm and 109.4 vs. 31.2 μg/hr/sq cm, respectively, p<0.10), a 2.3-fold longer Thalf (22.5 vs. 9.5 hr, respectively, p< 0.10) with a similar Tmax (1.4 vs. 0.75 hr, respectively).
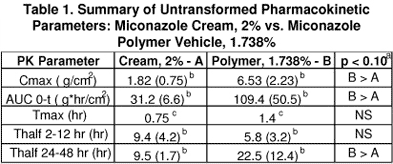
Table 1. Summary of Untransformed Pharmacokinetic Parameters: Miconazole Cream, 2% vs. Miconazole Polymer Vehicle, 1.738%
(a)paired t-test. bmean (SD), n=6. cmedian of n=6.
Stratum corneum drug content is presented as μg miconazole / sq cm adhesive disc normalized for control and dose. As shown in Table 2, the novel polymer vehicle (B) delivered more drug into human volar forearm stratum corneum in vivo than the cream (A) at 6 of the 10 time points investigated. Two to three-fold greater miconazole concentrations were produced with the polymer vehicle over the initial 0-12 hour uptake time interval than the semi-solid cream vehicle. Three to ten fold greater drug concentrations were achieved with the polymer vehicle (B) than the cream product (A) over the 24-48 hour time interval.
Figure 1 illustrates that the shape of the uptake/elimination profile of miconazole by the two products is strikingly different. The 2% semi-solid cream generates a typical rapid, but low uptake into stratum corneum followed by a slow elimination of the drug over time.
In contrast, the polymer vehicle produces a biphasic uptake/elimination profile; the initial uptake/elimination phase (0-12 hr) is maximal 2 hrs after application and may be associated with the co-delivery of the organic solvent phase of the vehicle.
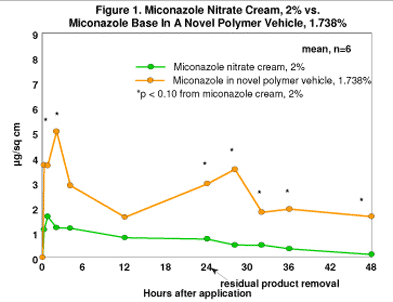
The latter uptake/elimination phase (12-48 hr) could be the result of variable drug product removal or an independent delivery mechanism associated with hydration of the skin site by the residual polymer vehicle and resolubilization of deposited crystalline drug on the skin surface.
Conclusion
Data support increased bioavailability of miconazole in human skin from the novel polymer vehicle compared to cream vehicle with regard to maximal stratum corneum drug content and duration of drug content 24 hours after residual product removal.
The ability to deliver more drug to skin may have great therapeutic effect and may require fewer applications leading to increased compliance and favorable treatment outcomes. Likewise, it may be possible to incorporate drugs at lower concentrations in the polymer vehicle to achieve effective topical drug therapy.
Acknowledgements
National Institutes of Health / National Institute of Allergy and Infectious Diseases. SBIR#R42AI041777. Sponsored by EpikeiaÒ Incorporated
Presented at: American Academy of Dermatology 64th Annual Meeting. San Francisco, CA, USA. March 3-7, 2006. Poster #P1114.