A NOVEL POLYMER-BASED SKIN BARRIER TO PREVENT AND TREAT IRRITANT CONTACT DERMATITIS
G. Siegel¹, BM Bhatt Ph.D.¹, Virginia Cardin¹, NJ Girard Ph.D., R.N., FAAN²,
¹Biomedical Development Corporation, San Antonio, TX, ²University of Texas Health Science Center at San Antonio,TX
Objective
This study investigated a polymer-based skin barrier to prevent and treat irritant contact reactions. The product is an acetone and polymer-based skin coating technology that is applied to the skin as a liquid that rapidly dries (~15 – 30 seconds) to form a thin film.
Irritant and allergic contact dermatitis affect approximately 10% of adults and hand eczema, in particular, represents one of the four most common occupationally-related diseases, accounting for substantial lost earning potential in otherwise healthy populations. Hand eczema is the most common occupational skin disease and occurs regularly in professions such as mechanics, textile workers, hair dressers, and especially healthcare workers.
Design
Sodium lauryl sulfate (SLS) was used as the model irritant. Six sites on the volar forearm were randomly assigned to the following treatments: 1. Control (untreated normal skin); 2. Negative Control (SLS applied only); 3. Barrier to Irritant Test (polymer coating applied, allowed to dry, then SLS applied); 4. Skin Barrier Function Recovery Test (SLS applied for 24 hours, then polymer coating applied); 5. Skin Barrier Function Recovery Test, Positive Control (SLS applied for 24 hours, then petrolatum applied); and 6. Polymer Effect on Normal Skin Test (polymer repeatedly applied daily to normal skin for 5 days, no SLS was applied).
Population, Sample, Setting
A nonrandom, convenience sample group of 26 adult male and female healthy volunteers aged 21-69 were enrolled in the study after signing informed consent. The 26 subjects were equally divided between whites and Hispanics, healthy by self-report, and had intact skin free of tattoos, scars, moles, vitiligo and keloids in the test areas. The 8-day study (6 visits) was conducted in a laboratory setting to control for environmental factors. Participants served as their own controls.
Methods
IRB approval, consent, and baseline measurements were completed. The designated treatment was randomly assigned and then applied to each of the sites described above.
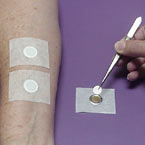
Sixty μl of an aqueous solution of 2.0% SLS was pipetted onto a layer of filter paper placed in each of several aluminum Finn Chambers® (Allerderm Laboratories, Petaluma, CA), which were affixed to the sites with hypoallergenic tape. On day 2, the Finn Chambers and barrier products were removed and all sites were scored for irritation and evaluated for TEWL. The study ended on Day 8 and final measurements were taken.
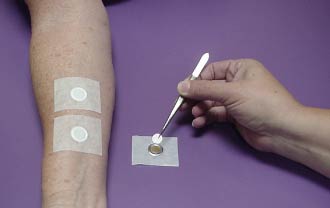
Figure 1. Filter paper saturated with SLS was placed into Finn Chambers and affixed to the test sites on the study subjects.
Outcome Variables
The two outcome variables were skin irritation and transepidermal water loss (TEWL). Irritation was evaluated visually and rank-scored. TEWL was determined using a Tewameter. TEWL is used as a quantitative means to assess skin barrier function. Damaged skin demonstrates increased water loss and thus, increased TEWL measurements.
Instrumentation
- Visual Scoring of Irritant Reactions: Each site was visually evaluated and scored daily for irritation according to the European Society of Contact Dermatitis guideline for clinical scoring of acute SLS irritant reactions1. Photos of the sites were taken on Days 2 and 8. Scoring was completed on a 0 (negative) to 4 (very strong/ caustic) scale.
- TEWL Measurements. Each site was evaluated daily for transepidermal water loss using a Tewameter TM210 (Courage & Khazaka, Cologne, Germany). TEWL measurements were made in a climatecontrolled room and followed recommended guidelines2.
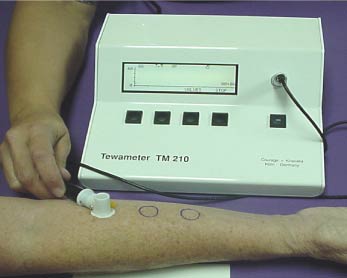
Figure 2. Transepidermal water loss recorded using Tewameter TM210
Results
Barrier to Irritant Test:
Sites receiving the polymer barrier and then SLS had lower TEWL levels than the SLS-only site (p<0.0001) at all time points, demonstrating the effectiveness of the polymer barrier to prevent irritant contact reactions. Visual evaluation scores confirmed the TEWL results, showing less irritation on the sites that received the polymer barrier than the SLS alone.
Skin Barrier Function Recovery Test:
At the final time point,TEWL measurements on the polymer-treated sites were significantly improved compared to the SLS-only sites (p < 0.027). There were no significant differences seen between the polymer-treated and the petrolatum-treated sites, suggesting that the polymer barrier was as effective as petrolatum at resolving the irritant contact reactions.
Polymer Effect on Normal Skin Test:
Repeated daily applications of the polymer barrier to normal skin for 5 days showed no significant differences between TEWL levels of normal, untreated skin. Thus, repeated application of the polymer barrier did not adversely affect normal skin.
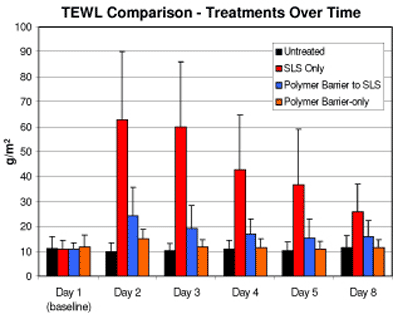
Figure 3. Barrier to Irritant Test: Transepidermal Water Loss at different timepoints for each treatment group

Figure 4. Barrier to Irritant Test Sites Day 2 and Day 8
SLS elicits a strong irritant response on the non-protected “SLS Only” test site. In contrast, the polymer skin barrier protects the skin from contact with the SLS irritant (Polymer Barrier + SLS site) and has a similar appearance to the untreated control. By Day 8, the irritant response has lessened considerably, but is still clearly visible. Note the small horizontal line of erythema on the lower-third of the “Polymer Barrier + SLS” treatment sites, which is likely the result of inadequate coverage of the polymer barrier on the skin.
Conclusion
The novel polymer skin product is an effective barrier to irritant contact dermatitis. When applied to irritated skin, the polymer coating improves skin barrier function as effectively as petrolatum. Applying the polymer barrier does not adversely affect normal skin.
References & Acknowledgement
1. Tupker RA, et. al. Guidelines on sodium lauryl sulfate (SLS) exposure tests. Contact Dermatitis. 1997; 37(2): 53-69.
2. Pinnagoda J, et. al. Guidelines for trans-epidermal water loss (TEWL) measurement. Contact Dermatitis. 1990; 22(3): 164-78.
Presented at: American Academy of Dermatology 64th Annual Meeting. San Francisco, CA, USA. March 3-7, 2006. Poster# P900
National Institutes of Health / National Institute of Allergy and Infectious Diseases. SBIR#R44AI045278.
Sponsored by Epikeia Incorporated