
SAFE, SIMPLE COST-EFFECTIVE MECHANISM TO CLEAN DENTAL UNIT WATERLINES
W.D. WARREN*¹, B.A. SANFORD², K.E. ESKEW², P.B.SIEGEL¹, AND G. SIEGEL¹
Biomedical Development Corporation1 and University of Texas Health Science Center2, San Antonio, TX
Introduction
Levels of bacteria detected in untreated dental unit waterlines (DUWLs) are typically μ1,000,000 colony forming units per milliliter (CFU/mL).1-3 Given that the acceptable standard for drinking water is [ 5 00 CFU/mL, DUWL water is generally considered unfit for human consumption. In 1995, the American Dental Association (ADA) established a year 2000 DUWL water quality goal of “no more than 200 CFU/mL of aerobic mesophilic heterotrophic bacteria at any point in time in the unfiltered output of the dental unit.”4-5
Daily treatment with Dentacide®, an iodine-based DUWL chemical treatment, has been clinically shown to satisfy the ADA’s goal of [ 200 CFU/mL by controlling DUWL biofilms.6-7 In addition, this product improves DUWL water quality without damaging dental unit water-circulating components.8 In this study, the iodine-based treatment was evaluated for biocompatibility and efficacy in a reduced frequency treatment protocol to determine the feasibility of a more costeffective maintenance protocol.
Safe
BIOCOMPATIBILITY TESTING METHODS:
Testing was performed by an independent laboratory, Northview Biosciences. All tests were performed according to federal guidelines for evaluating the biocompatibility of medical devices. Testing included assays for acute oral toxicity in rats, primary eye and dermal irritation in rabbits and dermal sensitization in guinea pigs. Extracts of polyurethane and silicon tubing exposed to the iodine-based chemical treatment were evaluated for cytotoxicity against mammalian cells in vitro.
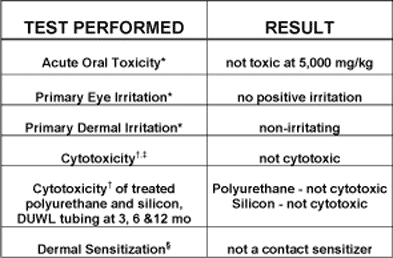
* per 16 CFR Part 1500.42, Consumer Product Safety Commission (CPSC), and Federal Hazardous Substances Act (FHSA) guidelines.
† U.S. Pharmacopeia 23 standardized test and compliant with International Standards Organization (ISO) Standard 10993-5.
‡ ASTM Standard F895-84.
§ ASTM Standard F720-81(reapproved 1992) and compliant with International Standards Organization (ISO) Standard 10993-5.

Use with an
independent
water reservoir
At the end of the day…

A. Remove remaining water

B. Add treatment to the water reservoir

C. Introduce the treatment into the waterlines and leave in overnight

D. Leave the water reservoir empty and store dry overnight
At the beginning of the day…

A. Fill the water reservoir with fresh water and flush lines
Cost-Effective
REDUCED TREATMENT PROTOCOL METHODS:
A reduced-frequency treatment, or maintenance protocol, was performed on university clinic dental units (>20 years old). Previously, all units had been treated daily with the iodine-based chemical agent to remove biofilm. Next, the units were either treated daily (control), twice weekly, or weekly for 8 weeks. Water samples (~5 mL) were collected weekly from the high-speed handpiece (HP) tubing of each unit and plated in triplicate on R2A agar containing 0.1% sodium thiosulfate. After incubation at 25 ! 2 )C for 7 days, total CFU/mL was determined. Data is expressed below as [ 2 00 CFU/mL (yellow) or >200 to <500 CFU/mL (blue).
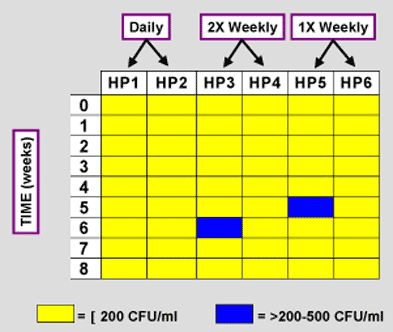
SUMMARY & CONCLUSIONS
Previous studies demonstrate that the iodine-based chemical treatment used in this study reduces DUWL contamination to a level consistent with the ADA’s year 2000 goal by controlling DUWL biofilms. The current study demonstrates that this DUWL treatment is non-toxic, nonirritating and non-sensitizing, and thus safe for patients and practitioners, should they ever be exposed. The success of this reducedfrequency treatment protocol demonstrates that once biofilm has been removed from the DUWL, the product can be used less frequently to maintain low bacterial counts. Less frequent use saves product, time, and therefore, money.
REFERENCES
- Miller CH. Microbes in dental unit water. CDA Journal 1996;24:47-52.
- Clappison RA. Priority one: decontamination of dental unit waterlines. Oral Health 1997;11-15.
- Barbeau J, et al. Multiparametric analysis of waterline contamination. Appl Environ Microbiol 1996;62:3954-59.
- Shearer BG. Biofilm and the dental office. JADA 1996;127;181- 89.
- ADA Online. American dental association statement on dental unit waterlines. Available at: http://www.ada.org/prac/position/lines.html.
- Sanford, BA, et al. Control of microbial biofilms in dental unit waterlines. J Dental Res 1999;78:262.
- Warren, WD, et al. Comparison of Dentacide and bleach in treated dental unit waterlines. J Dental Res 1999;78:262.
- Siegel, G, et al. Effect of Dentacide on dental unit watercirculating components. OSAP Annual Symposium 1999; #9919.
Funding was provided to Biomedical Development Corporation by The National Institute of Dental and Craniofacial Research at the NIH – SBIR No. 5R44 DE11221-03