TREATMENT OF KELOIDS AND HYPERTROPHIC SCARS WITH A NOVEL POLYMER-BASED SKIN COATING
G Siegel¹, BK Bhatt¹, JE Morrison¹, SD Bangert2, JH Miller¹, and AA Hebert¹
Biomedical Development Corp., San Antonio, TX¹and Dept. Dermatology, University of Texas Medical School-Houston, Houston, TX²
Objective
The purpose of this study was to evaluate a polymer-based film-forming technology as a treatment for mature hypertrophic scars and keloids in a 16-week clinical trial. The product is an acetone and polymer-based skin coating that is applied to the skin as a liquid that rapidly dries (~15 – 30 seconds) to form a thin film. The polymer formulation contains no silicone.
The primary efficacy endpoint was the Vancouver Scar Scale (VSS), Study participants also provided self-assessment of itching.
Background
Silicone sheeting is used as a non-invasive treatment for keloids and hypertrophic scars. However, silicone dressings often adhere poorly, can be difficult to affix to skin, can induce irritation on adjacent non-injured skin, and have inherent cosmetic deficiencies. This study was designed to evaluate the efficacy of a polymerbased skin coating to treat hypertrophic scars and keloids.
Methods
Forty volunteers having a mature (>7 months old) keloid or hypertrophic scar met inclusion/exclusion criteria and were randomized to either silicone sheeting or polymer coating treatment groups. Study participants self-administered the polymer coating treatment twice daily for the duration of the 16-week study period, and those randomized to the silicone sheeting group used the product consistent with instructions from the manufacturer. Scar measurements and subjective evaluation by the participants were completed at baseline and weeks 4, 8, and 16.
The Vancouver Scar Scale (Table 1), which provides a composite score for vascularity, pliability, and height, and an ordinal score for pigmentation. was used to assess scars. Study participants also provided self-assessment of itching (Table 2).
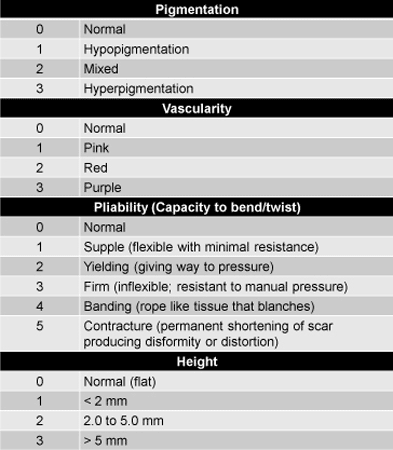
Table 1. Vancouver Scar Scale
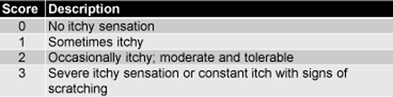
Table 2. Scale for Self-Assessment of Itching
Analysis
A repeated measures (pre-treatment/posttreatment) design was used for within and between group comparisons, with time as the group variable.. Wilcoxon Signed Rank Test and the Mann-Whitney U Test were used to explore within and between group comparisons where appropriate. Significance was ascribed to a p value ≤ 0.05.
Results
Seventeen subjects in the polymer coating group and 5 subjects in the silicone sheeting group completed the study and were considered evaluable. The high rate of drop-out in the silicone sheeting group was due primarily to complications from the treatment (skin irritation, difficulty of application, etc.), and disallowed treatment group comparisons.
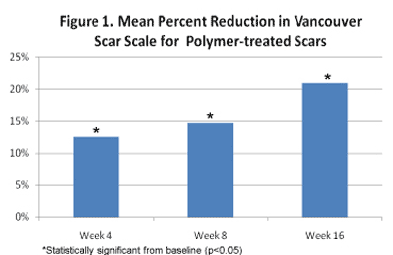
Subjects treated with the polymer coating showed a statistically significant difference in VSS index score from baseline at weeks 4 (p<0.006), 8 (p<0.001) and 16 (p<0.0001), demonstrating progressive improvement over the study period (Figure 1). Changes in scar pigmentation approached significance at week 4 (p<0.083) and continued treatment led to significant differences observed at week 16 (p<0.009).
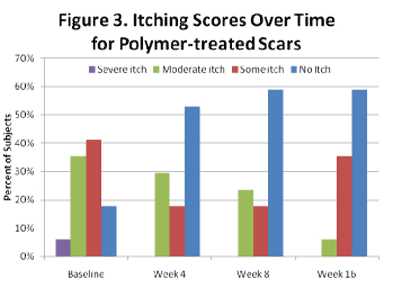
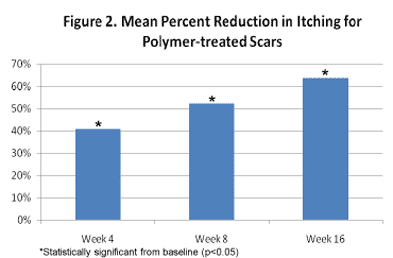
Significant and progressive improvement in itching sensation was reported at weeks 4 (p<0.034), 8 (p<0.017) and 16 (p<0.003) as shown in Figure 2. Notably, all individuals reported some degree of relief, with those reporting “no itching” increasing from 17.6% at baseline to 58.8% at the end of the study (Figure 3).
Discussion
Scars treated with the polymer coating showed significant and continual improvement over the four-month period. A disproportionately high number of patients dropped out of the study due to dissatisfaction and discomfort associated with the silicone sheeting. Similar dropout did not occur in those treated with the polymer coatings.
In order to minimize any effect of spontaneous scar resolution, only subjects with scars that were at least seven months old were enrolled in the study. In addition, there was no upper limitation placed on the age of the scar; as a result, many of the scars that were treated were several years old and reportedly refractory to previous modalities, including silicone sheeting. Better results may be expected in newer scars.
Conclusions
These novel polymer-based coatings are an effective treatment for mature keloids and hypertrophic scars.
Acknowledgements
National Institutes of Health / National Institute of Arthritis Musculoskeletal and Skin Diseases. SBIR#R43AR049972 .
Sponsored by Epikeia Incorporated and Skincerity Inc.
Presented at: American Academy of Dermatology 66th Annual Meeting. San Antonio, TX, USA. February 1-5, 2008. Poster #P3101.